More than 90 per cent of Australians could survive their cancer diagnosis within a decade under a comprehensive new framework developed by an alliance of expert researchers, doctors, patients, healthcare, tech and pharmaceutical leaders.
National Oncology Alliance (NOA) vision 20-30 report released today with an online forum which Rare Ovarian Cancer members attended.
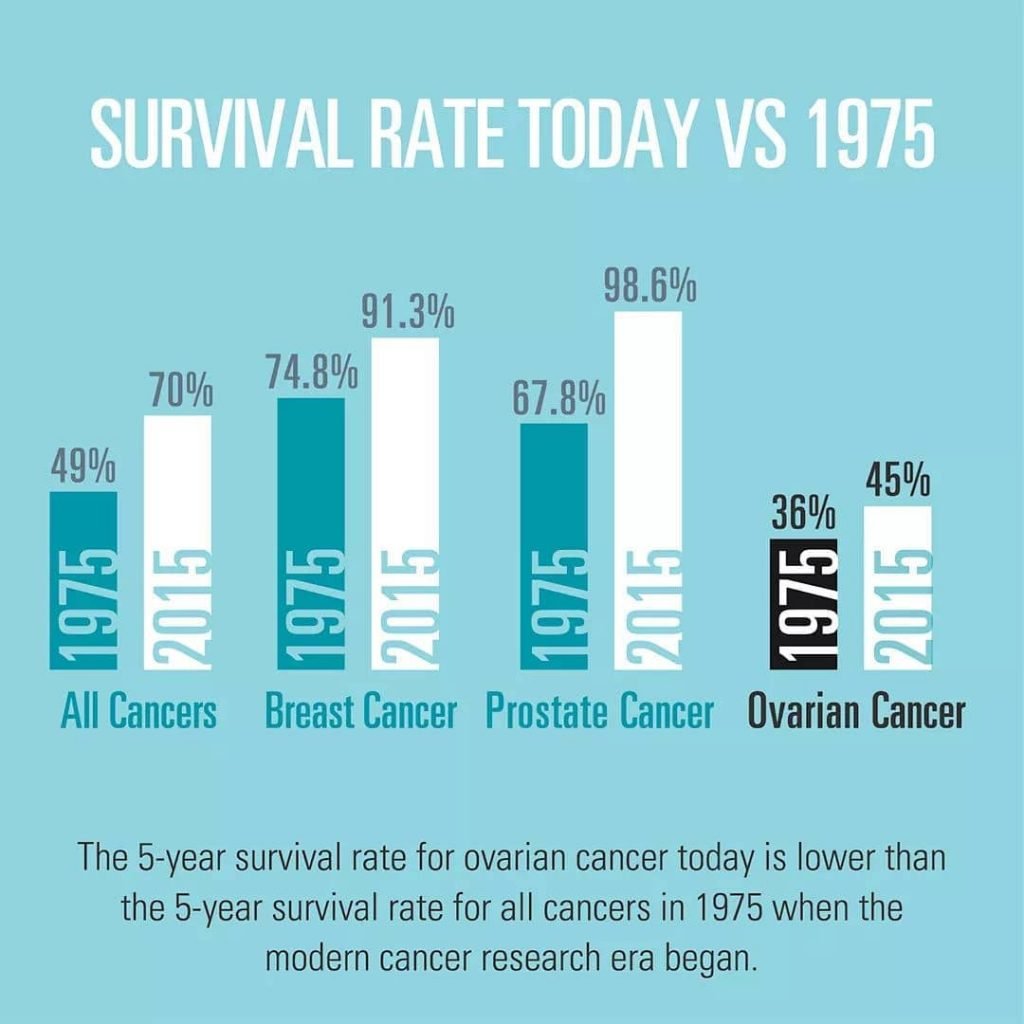
An ambitious but needed plan to aim for over 90 per cent survival rate for all cancers. For those diagnosed with Ovarian cancer a current 5-year survival of 46% has remained generally unchanged for over 30 years the survival rate would need to almost double in a 10 year time frame.
The National Oncology Alliance is calling on the government to urgently execute its plan – the Australian Cancer Futures Framework – to ensure Australia takes advantage of the life-saving advancements in cancer diagnostics and treatment.
More than 550,000 Australians are predicted to die from cancer in the next 10 years – 50,000 in 2020 alone, rising to 63,000 in 2030 if nothing changes, the alliance warns.
Alliance member and the chair of Rare Cancers Australia, Richard Vines, said Australia risks being left behind as the world moved on from a “one-size-fits-all” approach for treating cancer to precision, personalised medicine.
The six tenets of the Vision 2030 report
- Patient-centred cancer care
- Expanding clinical trials via telehealth to all patients regardless of geographical location
- Access to genomic sequencing to match patients with targeted therapies
- Integrate artificial intelligence within the healthcare system that could rapidly process immense amounts of data on cancer detection
- A centralised cancer registry that would aggregate comprehensive information on access to care, survival trends, variation in how patients are treated and genomic data
- Affordable, equitable and fast access to the best therapies and technologies for all cancer patients, including provisional access to drugs not subsidised on the Pharmaceutical Benefits Scheme.
